Payment service providers and online marketplaces should be aware of two drug ingredients that are making waves across e-commerce: ivermectin and fenbendazole. While these anthelmintic (antiparasitic) APIs have a legitimate use in human and animal medicine, the rise of their off-label use and unapproved forms is becoming a concern for consumer and card brand risk alike.
Keep reading to learn more about these problematic white label and “research use only” products that LegitScript is seeing across merchant portfolios.

May 4, 2026 | by Gabby Fisher
Antiparasitics: From Approved Use to Alternative Therapies
Antiparasitic drugs like fenbendazole and ivermectin have specific approved medical uses. Oral ivermectin is approved for human use by the FDA for antiparasitic treatment, and also has approved topical forms for the treatment of dermatological conditions like rosacea. Fenbendazole, however, is not approved for human use at all; instead, it is a common ingredient in approved animal deworming products.
Since the COVID-19 pandemic, ivermectin has been in the spotlight for reasons beyond its intended use. In early 2020, an initial laboratory study exhibited ivermectin’s inhibition of the replication of the COVID-19 virus in vitro. It was closely followed by a disputed secondary study which implied efficacy of ivermectin treatment in human cases of COVID-19. These early studies were quickly scrutinized and later withdrawn from scientific journals over fraudulent data and ethical concerns. However, this did not stop ivermectin from snowballing into a craze. Social media influencers touted it as a “miracle cure” and prescriptions issued for ivermectin rapidly increased. Today, its use is on the rise again as a panacea for various diseases, including cancer.
Similarly, fenbendazole has had its own share of misinformation-fueled hype. After an American cancer patient falsely claimed to have been cured by fenbendazole, it arose alongside ivermectin as a popular “alternative therapy” purported by independent media outlets and prominent public figures.
When Sales of Ivermectin and Fenbendazole Are Problematic
While legitimate pharmaceutical forms of these drugs exist, bad actors are finding sophisticated new ways to package and sell these ingredients for off-label use. LegitScript has observed ivermectin and fenbendazole sold as white label dietary supplements, unapproved drugs, and bulk API products labeled as “research chemicals.”
The following images are representative of the typical white label products that LegitScript has observed being sold across e-commerce.
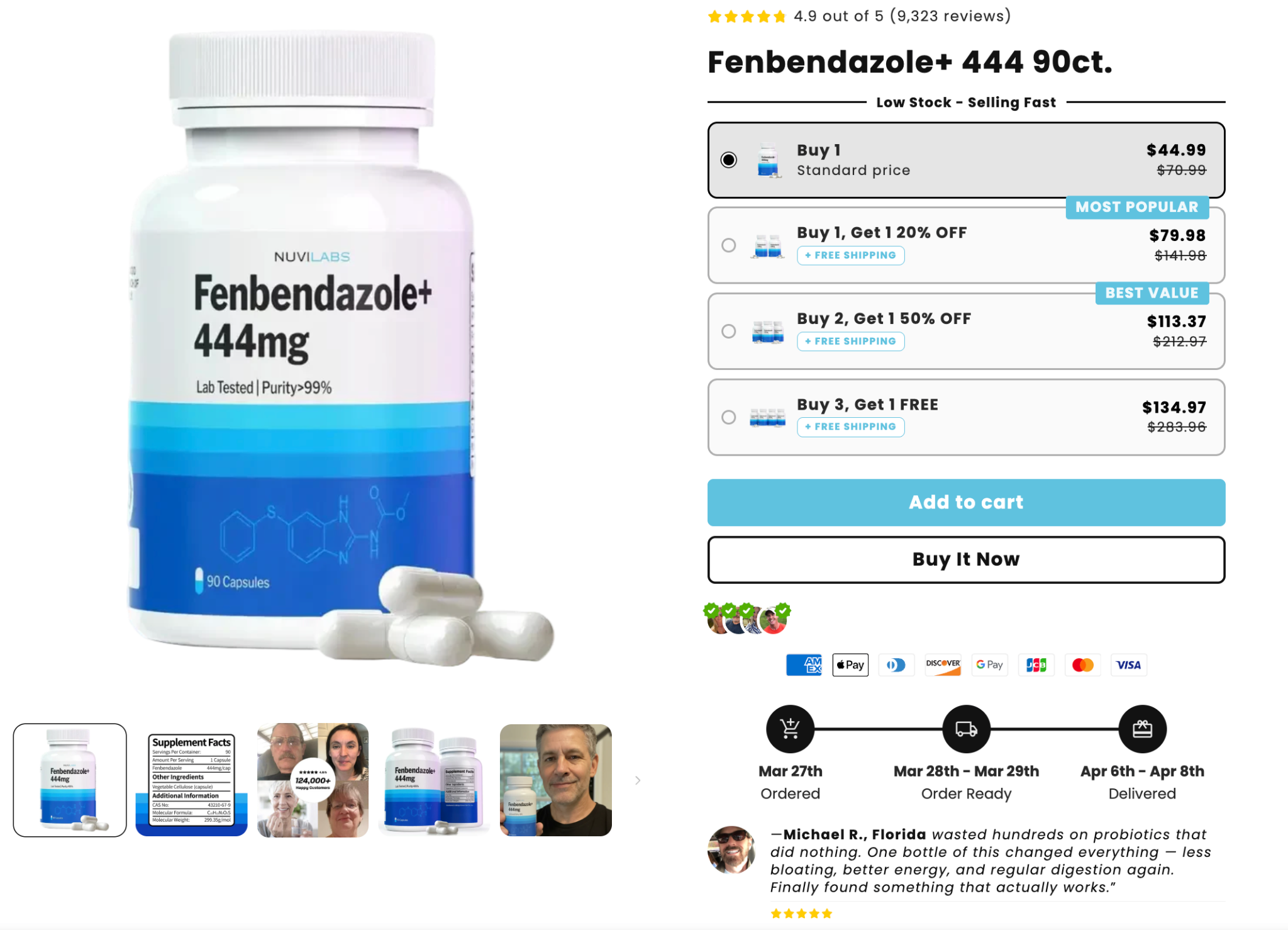
Fenbendazole marketed as a dietary supplement for human use
The image above features a merchant marketing fenbendazole as a dietary supplement for human use, with highlighted reviews emphasizing its probiotic effects. Fenbendazole is not approved for human use by the FDA, and has been cited for causing liver injury in multiple cases of self-administration and even death.
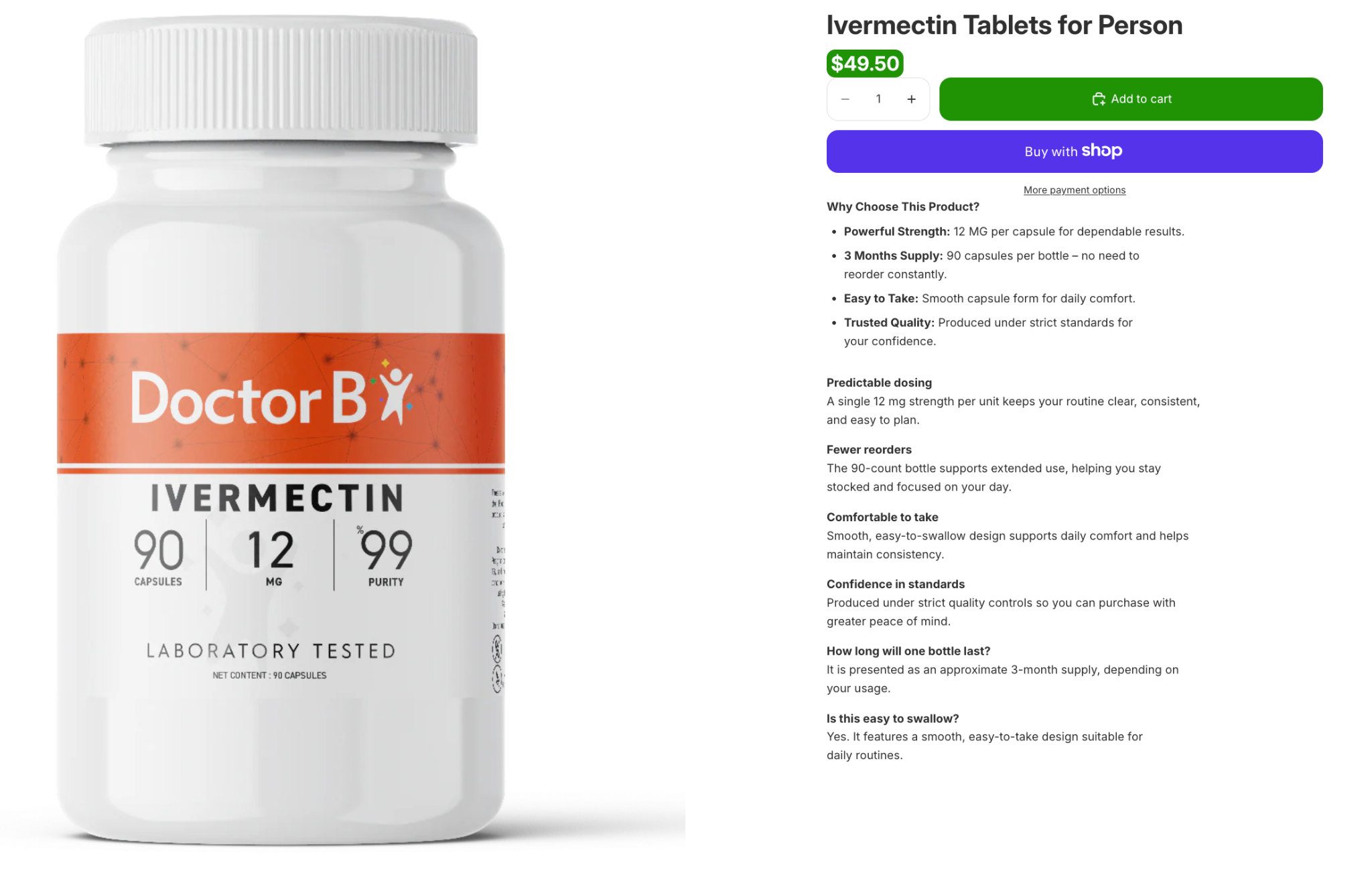
Ivermectin marketed online for off-label use
The image above shows a typical white label pharmaceutical product that contains ivermectin (oral), which is a prescription-only active pharmaceutical ingredient in many jurisdictions, including the United States. This product is not only unapproved, but the merchant also lacks proper prescription verification safeguards.
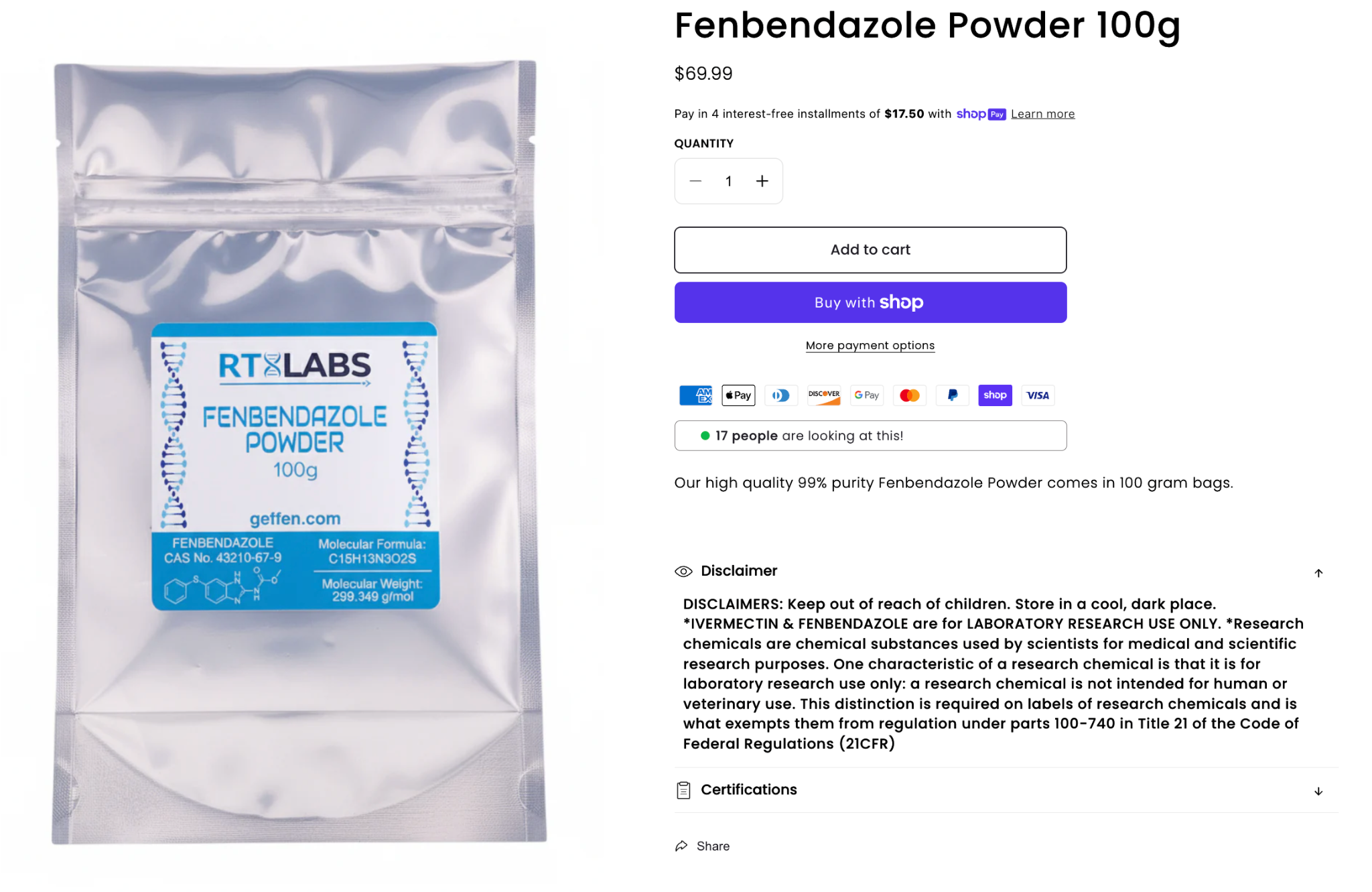
Fenbendazole powder sold for "research use"
Additionally, LegitScript has observed merchants selling these APIs under the guise of research chemicals, mirroring the trends we are seeing across the healthcare e-commerce space with other products like peptides. The image above features a merchant listing that offers bulk fenbendazole powder, with no safeguards to ensure that products are only purchased by professionals for research purposes.
What Are the Risks of Ivermectin and Fenbendazole?
Even approved drugs may have adverse side effects, which is why the FDA recommends exercising caution when using approved drugs for off-label use. This risk increases significantly with white label products and research chemicals that are not approved for human consumption. Because these products are unapproved, they inherently present a risk of being unsafe, ineffective, and inconsistent in quality. As of early 2026, the FDA appears to have intensified its focus on misleading advertising for unapproved drug products, issuing dozens of warning letters to telehealth companies for similar deceptive marketing.
Besides consumer health risk, these products present immediate regulatory and reputational risk. Because these “white label” drugs are unapproved and sold without proper oversight, they are risky transactions. Card brands are aggressively cracking down on these products and issuing substantial fines to acquirers whose portfolios contain merchants selling them.
Safeguard Your Merchant Portfolio With Leading Industry Intelligence
LegitScript’s Merchant Monitoring solutions are tailored to identify unapproved products and services in more than 60 high-risk verticals. Our combination of AI-powered technology and human review offers both speed and accuracy so that you can stop problematic merchant behavior before card brand fines hit. Our analysts are specially trained in evaluating the risk level of complex drug products so that you can grow your portfolio with merchants who are in compliance while taking against those who are not.
Protect your merchant portfolio from the rise of unapproved antiparasitics and other problematic products. Contact us today.



