Dietary supplements containing peptides are popping up across e-commerce. These products are often marketed for building muscle, promoting longevity, or healing from injury. These claims sound compelling but many peptide ingredients sold in supplement form raise regulatory concerns.
Keep reading to learn more about the risks of peptides in supplements, and then download our recent guide Understanding Peptides.
February 9, 2026 | by Gabby Fisher
The Food and Drug Administration (FDA) has flagged safety concerns regarding certain peptides, and has not approved many of them for human use. When merchants sell supplements containing unapproved peptide ingredients, payment industry stakeholders and e-commerce marketplaces can face added regulatory scrutiny and potential card brand enforcement. Below we outline what you need to know about this emerging threat.
What Is a Peptide?
Peptides are a short chain of amino acids, which are the building blocks of proteins; their distinct therapeutic properties present exciting possibilities for disease treatment. In medicine, some peptides are widely studied and used in approved drugs. For example, semaglutide and tirzepatide are available as diabetes treatments in FDA-approved prescription products such as Ozempic and Mounjaro.
However, many other peptides sold online have not been evaluated to the same standard for safety, effectiveness, or appropriate use. Some are marketed for “research use only,” while others are presented as wellness products despite lacking approval for human use in the US. In some cases, the FDA has identified specific peptides as presenting safety concerns, adding them to their dangerous substances list.
Evolving Forms: From Invasive Injections to Convenient Capsules
Unapproved peptides have long been a monitoring focus for LegitScript because they often appear as injectable products and are commonly marketed as “research use only.” As FDA and card brand enforcement increases, merchants and manufacturers are finding new ways to package and market them in an effort to evade detection, including selling peptides in oral supplement formats such as capsules, powders, and gummies.
These supplements offer consumers a convenient way to ingest peptides, and allows merchants to operate under a highly marketable and financially lucrative guise of efficacy. While certain dietary supplement ingredients may have certain health benefits, many are marketed with unsubstantiated claims, which are often heavily promoted by both celebrities and social media influencers alike. Among these are peptides like BPC-157 and Thymosin beta-4 fragment; both appear on the FDA’s dangerous substances list and have recently featured in merchant portfolios in dietary supplement form.
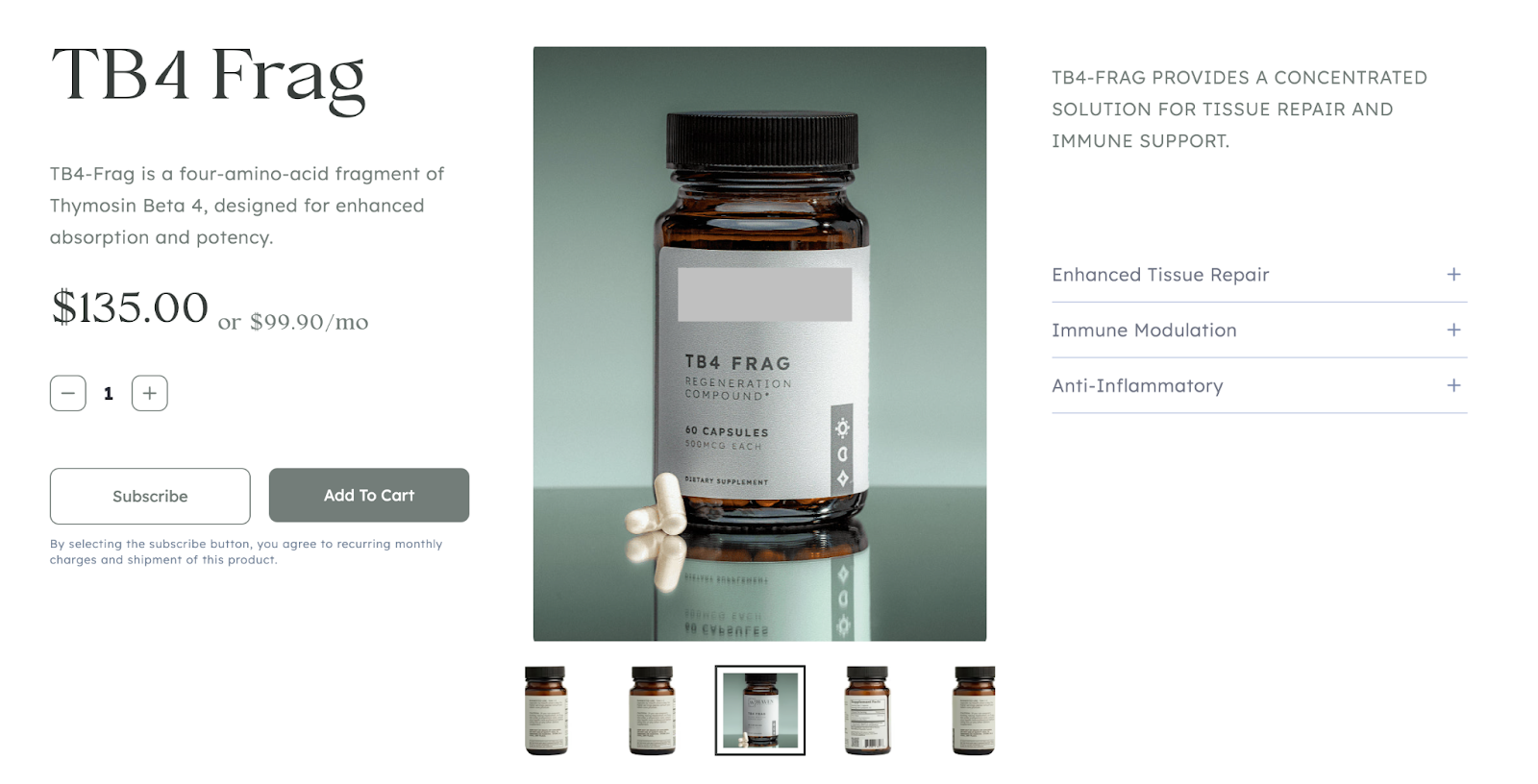
Tβ4 Frag is often marketed for wound healing, anti-inflammation, and organ repair.

BPC-157 is often marketed for bone and joint healing and for increasing athletic performance.
These supplements are often promoted for muscle and tissue repair, anti-inflammatory effects, and enhanced immune response. However, the benefits may not outweigh the risks. Their unapproved status presents not just health and safety concerns for consumers, but financial risk for merchants and payment partners who facilitate the sale of these products.
Mitigating Risk: LegitScript Stays on Top of Peptides
Despite their unapproved status, peptide-containing supplements are often packaged in ways that suggest legitimacy, and some merchants may offer them in good faith without understanding their regulatory status. Card networks and regulators may still treat these products as high risk, especially when listings imply drug-like effects or include unapproved ingredients.
Until the FDA clearly allows specific peptides for use in dietary supplements, peptide-containing products are likely to remain a source of compliance and enforcement risk. LegitScript helps reduce exposure by monitoring regulatory guidelines around high-risk products across merchant portfolios. Our technology and analysts detect peptide-related risk signals, including unapproved ingredients and problematic health claims, and provide clear reporting that supports quicker action.
Learn More
Want to protect your merchant portfolio from card brand scrutiny related to unapproved peptides and other high-risk verticals? Learn more about LegitScript’s Merchant Monitoring program and how our risk solutions support ongoing compliance.
Understanding Peptides
A Q&A Guide for Payment Processors and Online Platforms
In this in-depth guide, you’ll get introduced to the peptides industry, gaining a better understanding of how it works and learning why there has been a recent surge of interest in these experimental products.




